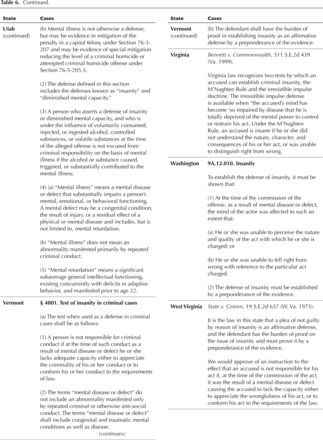
Methane Production Essay - 4039 Words - StudyMode.
Methane Recovery from Landfills Utilization as a Potential Energy Source and Impact on Reduction of Green House Gasses According to The Conference Board of Canada, current Canadian municipal solid waste (MSW) generation levels are approximately 30 million tonnes per year, with a rate of 894 kg per capita, 67 percent of which is landfilled.(Jones L.et al.
The primary compositions of natural gas are methane (CH4), ethane and propane. Natural gas is mainly used to fuel electric power generators, the raw material for making consumer products from plastics and heats buildings. This paper explores the advantages and problems associated with extraction, processing, transportation and use of natural gas.
Methane Formation Methane formation beneath permafrost is created by three different processes. To begin, methane can be made through the thermal decomposition of organic matter, known as being produced thermogenically. Furthermore, methane can also be produced abiogenically and bacteriogenically (Stotler et al., 2010).
Methane, the primary component of natural gas, is a powerful, short-lived greenhouse gas. It is more than 100 times more potent at trapping energy than carbon dioxide (CO2), the principal contributor to man-made climate change. When considering its conversion to carbon dioxide over time its impact on an integrated weight basis is 84 times more.
Methane is a key precursor gas of the harmful air pollutant, tropospheric ozone. Globally, increased methane emissions are responsible for half of the observed rise in tropospheric ozone levels. While methane does not cause direct harm to human health or crop production, ozone is responsible for about 1 million premature respiratory deaths.
Other posts on the site.
Methane, colorless odorless gas that occurs abundantly in nature and as a product of certain human activities. Methane is the simplest member of the paraffin series of hydrocarbons and is among the most potent of the greenhouse gases. Learn more about the properties and uses of methane in this article.
Important Initiator For Methane Combustion Biology Essay A burning reaction can be defined as a fast chemical reaction between a fuel and an oxidant that releases heat. Traditionally, the fuels used in the bulk of practical burning applications have been hydrocarbons, chiefly due to their high energy denseness and easy handiness (till late).





/1905_Wallace_A237.1_135.jpg)